We continue our series with some of the most strange and unknown animals on earth. In Strangers in the animal tree (Pt1) we got to known two monotypic phyla with mouth issues: Ciclyophorans, who are only found in the mouth of some common lobsters and Micrognathozoans, which have one of the most complex mandibles on Earth, despite being just some millimeters in size!
In this article we take a look at Placozoans and Orthonectides, two clades which seem to belong to an era when the separation between animals and fungi wasn’t clear …Or they are just the result of a parasitic life/ex-life, something more prosaic but always interesting, nonetheless.
Placozoa, the 3 layer-only animals
The non-bilaterian animals (which means their body cannot be separated into equal halves, amongst other characteristics) are essential to understand the first steps in the evolution of animals and have been targeted to clarify the origin of neurons, muscles and other cell types. Their study is therefore vital if we wish to understand the evolution of animals and also ourselves.
Non-bilaterians have somewhat amazing characteristics: Sponges (Porifera) are able to reconstruct their bodies after being destroyed, Jellyfishes (Cnidaria) have cnidocites (unique harpoon-like cells) and Comb Jellyfishes (Ctenophora) are the largest animals to be able to move only with cilia (which also creates their disco-like colorations), just to name a few of them.
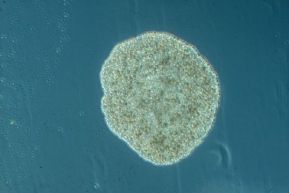
And last but not least, we find Placozoans, whose main characteristic is… having no main characteristic. What you see in the image above is a fully, complete, functional animal. Discovered in 1883 in an aquarium of Austria, the specie called Trichoplax adhaerens is the only species known of Placozoa, and unique in its own right.
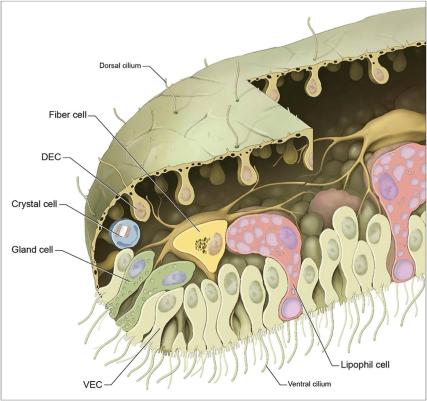
Although I am normally against the complex-simple dichotomy, it’s clear that Trichoplax, with a 2-3 mm large and 25 µm width body, only three layers of cells (check the image by Smith et al.) and 6 types of cells, is not a difficult organism to understand. Despite this simplicity, though, this little animal still guards many secrets to be unveiled.
First of all, let’s take a look at its overall shape. The upper layers have small cilia, while the basal ones have microvilli (extensions of the cells that resemble tentacles) that are used for movement. The middle layer is composed mainly of cells separated by septa (“walls” that divide cells). Interestingly, septa is a type of cell structure commonly found in fungi, but it only appears in the animal kingdom in Placozoans and some sponges. And this is only the beginning of Placozoans’ mysteries.
As an example, despite having been kept into aquariums for decades, its sexual reproduction is still unknown. Until now, it has only been observed that once in a while, two parts of the organism start to move in opposite directions, which ends up splitting up the animal and giving two Placozoan daughters! Other types of asexual reproduction have been observed, such as budding, but no trace of sexual reproduction has been found.
Another interesting pattern is its feeding behavior. According to some studies, the search for food and its capture is a complex procedure in these animals, which could only be explained if a cell-connection system existed. This would be usually the work of neurons but… Placozoans lack neurons.
And not only neurons, they lack many cell types that Sponges or Jellyfish do have, even though according to current classifications, Placozoans are newer in the evolutionary history of animals than these phyla. Basically, this means that Placozoans are not a group connecting fungi with animals or a primitive animal. In fact, Placozoans have many genes that code for body structures that aren’t being expressed! Furthermore, 85% of their genes are also found in other groups, which means despite their weirdness in phenotype, their genotype is not particular at all.
To sum up, everything seems to support the hypothesis that Placozoans used to be parasitic animals, losing all previous unnecessary traits, such as neurons… However, we don’t find these animals as parasitic today. So… Have they returned to a non-parasitic life? If that’s the case, why?
In Strangers in the animal tree (P1) I promised I would talk about one of the “newest incorporations” in the animal tree of life, but I will leave this for future issues (where you will understand why I wrote newest incorporations in brackets ) and talk instead about…
Orthonectida, the amorphous king
With 20 species, we enter the biggest group of this phyla series. But don’t expect anything “usual”, as Orthonectides are in pair with Placozoans in terms of simplicity and amount of mysteries around them.
Obviously, with 20 species, Orthonectides are far from being monotypic (they contain at least four potential families and around six genre, such as Rhopalura, Intoshia or Pelmatosphaera). As for their phylogenetic position, it seems they could be related to Annelida or Mollusca, but that doesn’t tell us much abouth thier morphology, as it clearly deviates from the common pattern of these two phyla.
Orthonectides are worm-like animals comprised of two layers of cells with cilia and gonads, which is a structure even simpler than the one of Placozoans. They are completely parasitic and infect marine animals from completely different phyla, ranging from Platyhelminthes (flat worms), Cephalopods (Octopuses and Squids) or Echinoderms (Sea Urchins, Sea stars…).
A parasitic life normally leads to the ultimate reduction of the animal, as we have seen in Placozoans. Rhombozoans are another group of parasitic animals which greatly resemble Orthonectides (in fact they were placed together as Mesozoans for a long time) but are completely different, so their similar shape is only the result of having the same life-style.
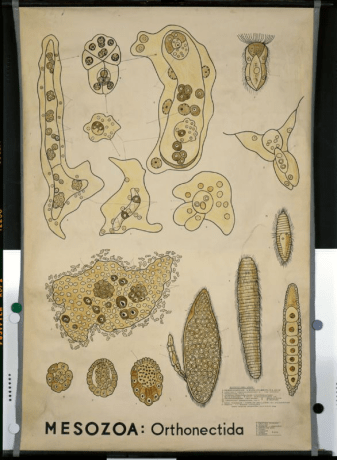
The life cycle of Orthonectida is different from the Rombozoan one, and as always, rather complex (not as complex as the one in Cycliophora, luckily). Orthonectides have a free swimming stage (can be seen in the first image from the article) where the female is as twice as big as the male and a parasitic stage, where their body transforms completely into an amorphous being made up of cell clusters, the plasmodium (can be seen in the image of Hyman 1940), which takes its name from a similar structure found in fungi. This structure can develop to great sizes inside the host, heavily damaging it.
At some point, the plasmodium breaks into pieces, each one generating an adult individual. These adult stages are free ciliated swimming individuals either male or female depending on their plasmodium parent. The male attaches to the female and their content gets fused. Fertilization occurs and new larvae develop into the female’s body. They afterwards find a new host, getting attached to it, beginning a new cycle. Truly amazing.
In the next issue….
We will take a look at Phoronida, the phylum that looks like corals (but in fact are not) and Dendrogramma, the “phylum” that shook the scientific community on 2014 and one of my personal favourite animals.
If you enjoy our articles, follow us on Twitter and Facebook! Also follow the author on twitter!
Bibliography
Hanelt, B., Van Schyndel, D., Adema, C. M., Lewis, L. A., & Loker, E. S. (1996). The phylogenetic position of Rhopalura ophiocomae (Orthonectida) based on 18S ribosomal DNA sequence analysis.Molecular biology and evolution, 13(9), 1187-1191.
Pawlowski, J., Montoya-Burgos, J. I., Fahrni, J. F., Wüest, J., & Zaninetti, L. (1996). Origin of the Mesozoa inferred from 18S rRNA gene sequences.Molecular biology and evolution, 13(8), 1128-1132.
Robert D. Barnes (1982). Invertebrate Zoology. Philadelphia, PA: Holt-Saunders International. pp. 247–248.
Slyusarev, G. S., & Miller, D. M. (1998). Fine structure of the mature plasmodium of Intoshia variabili (Phylum Orthonectida), a parasite of the platyhelminth Macrorhynchus crocea. Acta Zoologica, 79(4), 319-327.
Smith, C. L., Varoqueaux, F., Kittelmann, M., Azzam, R. N., Cooper, B., Winters, C. A., … & Reese, T. S. (2014). Novel cell types, neurosecretory cells, and body plan of the early-diverging metazoan Trichoplax adhaerens. Current Biology, 24(14), 1565-1572.
Smith, C. L., Pivovarova, N., & Reese, T. S. (2015). Coordinated feeding behavior in Trichoplax, an animal without synapses. PloS one, 10(9), e0136098.

2 Comments Add yours