
Around 500-million-years ago, all macroscopic life was still living underwater. Animals were still relatively young, and plants still had tens of millions of years to evolve and claim the land. You have probably heard about this time: the Cambrian period. The Cambrian is probably one of most popular paleontological eras without dinosaurs (in fact, almost 300 million years before they first appeared), highly popularized thanks to palaeontologist Stephen Jay Gould’s book Wonderful Life, and the general bizarreness of the animals of that period.
Among the many bizarre animal forms from the Cambrian (check the first image), one group may look more familiar to the untrained eye: shrimps. These “shrimps”, though, have something unique: they bear a carapace on top of their head and thorax. When examined more closely, we see that these are not really “shrimps” but rather a diverse series of shield-bearing shrimp-like animals: the Cambrian bivalved arthropods.
What were these ‘bivalved arthropods’? And what role did they play in the evolution of one of the most successful animal groups in the history of this planet?
The Cambrian: the time when animal life “exploded”
The Cambrian period was a particularly interesting time in the history of Earth. Life had appeared thousands of millions of years before the Cambrian and had since diverged into the main groups of living organisms we know of (bacteria, algae, fungi…). However, one of the most significant groups of living organisms still had to make a presence into the world: animals.
Around the XIX Century, Cambrian fossils were discovered and put into a geological context for the first time. These were some of the oldest fossil-bearing rocks known, and thus, the earliest clues of animal life. Darwin and Wallace welcomed these fossils as part of their theory of evolution by natural selection, but some of their contemporaries used them to disprove that same theory. The problem? The Cambrian animals were too complex! How was it possible that earliest animals already had shells, segmentation, eyes or exoskeletons? If evolution was slow, as Darwin and Wallace had suggested, how did these animals evolve these traits? This couldn’t be anything but an act of divine creation. Darwin, instead, had this to say:
“it is indisputable that before the lowest Cambrian stratum was deposited long periods elapsed, as long as, or probably far longer than, the whole interval from the Cambrian age to the present day; and that during these vast periods the world swarmed with living creatures.”
On the Origin of Species, 1859
Darwin, then, reasoned that there must be fossil-bearing rocks even older than Cambrian ones. It was either this, or evolution didn’t work exactly as Darwin envisioned. Both turned out to be true.
It took multiple misunderstood and unaware geologists, palaeontologists and fossil amateurs, as well as almost 100 years since Darwin’s statement until the 600 million-year-old fossils of Ediacara were discovered in the 1960s. While there is still some debate on what some of these fossils are, many are regarded as some of the first animals we know of.


In a sense, then, Darwin was right, and the Cambrian is just but another stepping stone in the evolution of animals. Case solved? Not yet. The animals from Ediacara look like what we would expect the first animals to look like: worm-like, flattened, soft-bodied creatures feeding on bacterial mats or filtering water. Jellyfishes and sponges were some of the most complex creatures of that time. However, there are two things about them that we did not expect:
–Most Ediacaran animals are not classifiable in our current animal groups. In fact, many show features in their general body structure that are not found among extant animals. While our animals are amorph (sponges), radially symmetrical (jellyfishes) or bilaterally symmetrical (all other animals), for example, some Ediacarans have tri-radially symmetrical bodies (as we saw in Strangers in the animal tree (FINALE)).
–There is no smooth transition between the features shown in Ediacaran animals and features in Cambrian animals. While the Ediacaran animals are very interesting and unique by themselves, the Cambrian animals are the ones that show many of the features that current animals have: eyes, antennae, mussels, shells, fins… These were not present in the Ediacara, meaning that in only 100 million years, all of these features had to evolve. This is not a lot of time, in evolutionary terms. In fact, one can say that evolution was fast, experimental, or that there was a ‘Cambrian Explosion‘.
Studying the Cambrian, then, is not only studying ancient animals and ecosystems, but also answering one deep question: why was evolution so fast and creative during that time? And what does this tells us about how evolution works?
A quick introduction to Bivalved Arthropods.
Today, arthropods are not only the animal group with the highest number of species, but also with the highest disparity. Disparity measures how different are two objects from each other and is a very interesting metric to understand what are the limits of evolution. Arthropods may have originated among the Ediacaran rarities, but is is only in the Cambrian that we see both animals clearly recognizable as arthropods and the origin of the main arthropod groups. Species such as Mollisonia or Habelia are probably the ancestors of what we know today as chelicerates (spiders, horseshoe crabs…). Trilobites also appear in the Cambrian for the first time. And of course, there are the first mandibulates, a group that combines myriapods (centipedes, millipedes…), crustaceans (crabs, shrimps…) and insects (bees, ants…). And not only that, the Cambrian shows a majestic diversity of other arthropod groups which lineages went extinct: the horned Marrella, the scavenging multisegmented Fuxianhuia or the three-digited clawed ‘shrimp ‘ with whips Yawunik are just some of examples of that diversity.
One type of fossil, though, is specially common in the Cambrian: carapaces. But these carapaces do not belong to shelled animals like bivalves or brachiopods, but arthropods. All arthropods have their bodies covered by small plaques of chitin, an impermeable and somewhat sturdy structure. In some arthropods, though, these plaques grow and envelope the body, thus, creating a “shield” or a carapace (for an extant counterpart, the “head” of a shrimp can be considered a carapace). Bivalved arthropods, then, are a group of arthropods having wide carapaces that cover their head, thorax and can even cover their entire body. The carapace, though, is but a convergent structure, meaning that widely different arthropods evolved a carapace independently.
But why are bivalved arthropods important? Why is this series dedicated to them?
- Bivalved arthropods are an evolutionary puzzle: there are still many questions regarding which role every group of bivalved arthropods played in the early evolution of arthropods. Things have improved a lot in the last decade, but multiple pieces of the puzzle are still missing or in debate.
- Bivalved arthropods may be key to understand the origin of mandibulates. Mandibulates have since spread through sea and land, evolving into a myriad of forms and many different ecological niches (predators, filter-feeders…). Were bivalved arthropods behind this success?
- New bivalved arthropods are showing diverse, unique features. The Cambrian is rich in oddities, but most bivalved arthropods look similar to phyllocarids, a group of ‘shielded shrimps’ that still exists to date. Recent discoveries are showing that bivalved arthropods are quite different among each other (high disparity), and many have features, such as the ventrally extended spine of Fibulacaris, or the double shield of Pakucaris that were lost in time, and we don’t even see among the myriad of mandibulate species.
- Bivalved arthropods were ecologically diverse, and had complex behaviours. Do you know that many shrimps lay their eggs on their legs or carapace, protecting them? Well, bivalved arthropods like Waptia did it earlier. And that some lobster create ‘conga lines’ to migrate as a group? Bivalved arthropods like Synophalos also did it earlier.
Slideshow: different bivalved arthropods mentioned: Fibulacaris, and odaraiid with its ventral spine (C) ROM; Pakucaris, an odaraiid with its second shield (C) ROM; Synophalos, a hymenocarine, making conga-lines (from Hou et al., 2009); Waptia, a hymenocarine, holding eggs in its carapace (C) ROM.
Main bivalved arthropod groups
In more than 100 years of Cambrian research, several groups of arthropods have been called ‘bivalved arthropods’. The definition on what is a ‘bivalved arthropod’, then, is completely fuzzy. The following is a list of all those groups, that wear a carapace and were called, at some point ‘bivalved‘.
Bradoriids:
Tiny and abundant, the bradoriids were probably close to the bottom of the Cambrian food chain. Bradoriids were able to encapsulate their body inside their carapace, and had notches on their carapace that accommodated their eyes. Bradoriids like Kunmingella are known to protect their eggs. Recent advances using computerized tomography are starting to reveal what lies behind the carapace.
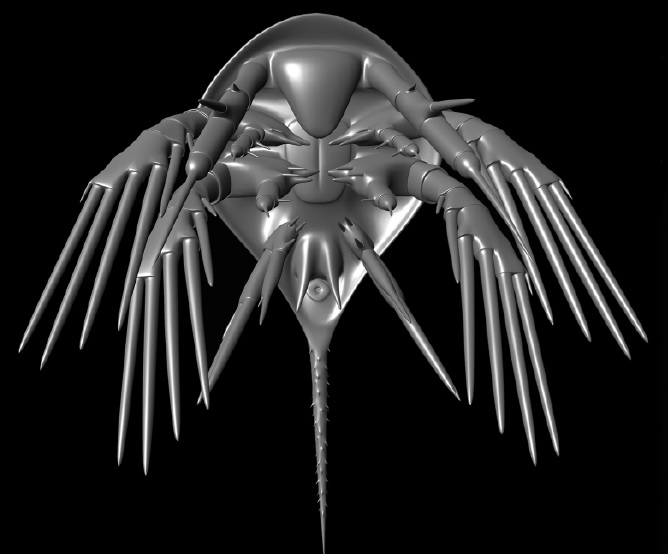
‘Orsten crustaceomorphs’:
Larvae of unknown adults, part of the plankton, these are some of the best fossils in the world. Smaller than one millimetre, these animals were preserved through phosphatization, and show detailed structures, such as hairs and spines. As larvae, they are difficult to place in the arthropod tree of life, but these may be some of the first crustaceans we know of.

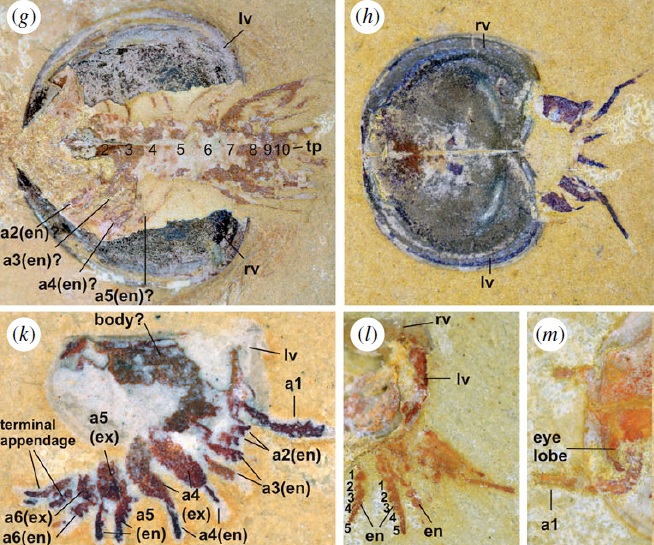
Phosphatocopids
Similar to bradoriids or the extant ostracodes, the body of phosphatocopids is enclosed in a carapace. Microscopic specimens in ‘Orsten’ deposits show that their body is very different from bradoriids, and show more derived features, such as mandibles. Phosphatocopids may be the larvae of some of the first crustacean groups, like barnacles, but their position is also in discussion.

Isoxyids
Isoxyids are characterized by their big multisegmented claws and enormous eyes. They are believed to be some of the first ‘euarthropods’, meaning some of the first arthropods to have birramous legs. Isoxys probably inhabited the upper layers of the water column, and was able to all across the Cambrian tropical seas. The other two genera: Surusicaris and Erratus, are quite different, and only known from a few specimens.

Hymenocarines
Hymenocarines are not the most diverse group, but the most disparate group of bivalved arthropods. Their carapace covers only half or three-quarters of their body, and the shape of their body, legs and tail vary greatly among species. They had different head and trunk conformations, as well as a wide variety of feeding strategies and behaviours. The discovery of mandibles in several species suggests they may be the first mandibulates.

‘Odaraiids’
The ‘odaraiids’ have highly multisegmented body and reduced or absent antennae and were mostly filter-feeders. Many have never-seen-before carapace shapes. Given their small size, the anatomy of their head is poorly known. Due to this, while some think that they show ‘primitive’ features, that would them place them close to the first euarthropods (like isoxyiids), others think they are just a group of hymenocarines.
Figure source from top to bottom: Kunmingella in Hou et al., 2010; Sandtorpia in Haug et al., 2010; Vestrogothia in Maas et al., 2005; Isoxys in Legg et al., 2013; Tokummia in Aria & Caron 2015; Fibulacaris in Izquierdo-López & Caron, 2019.
What do you think about bivalved arthropods? If you want to know more, we will cover some of these groups in the nearly feature in more detail. In the meantime, if you liked this post, please follow us on the following social media!
References
Cambrian & Cambrian Explosion
Aria, C. (2020). Macroevolutionary patterns of body plan canalization in euarthropods. Paleobiology, 46(4), 569-593.
Briggs, D. E., Fortey, R. A., & Wills, M. A. (1992). Morphological disparity in the Cambrian. Science, 256(5064), 1670-1673.
Darroch, S. A., Smith, E. F., Laflamme, M., & Erwin, D. H. (2018). Ediacaran extinction and Cambrian explosion. Trends in ecology & evolution, 33(9), 653-663.
Erwin, D. H., Laflamme, M., Tweedt, S. M., Sperling, E. A., Pisani, D., & Peterson, K. J. (2011). The Cambrian conundrum: early divergence and later ecological success in the early history of animals. science, 334(6059), 1091-1097.
Lee, M. S., Soubrier, J., & Edgecombe, G. D. (2013). Rates of phenotypic and genomic evolution during the Cambrian explosion. Current Biology, 23(19), 1889-1895.
Marshall, C. R. (2006). Explaining the Cambrian “explosion” of animals. Annu. Rev. Earth Planet. Sci., 34, 355-384.
Bradoriids
Hou, X., Williams, M., Siveter, D. J., Siveter, D. J., Aldridge, R. J., & Sansom, R. S. (2010). Soft-part anatomy of the Early Cambrian bivalved arthropods Kunyangella and Kunmingella: significance for the phylogenetic relationships of Bradoriida. Proceedings of the Royal Society B: Biological Sciences, 277(1689), 1835-1841.
Zhai, D., Williams, M., Siveter, D. J., Harvey, T. H., Sansom, R. S., Gabbott, S. E., … & Hou, X. (2019). Variation in appendages in early Cambrian bradoriids reveals a wide range of body plans in stem-euarthropods. Communications biology, 2(1), 1-6.
‘Orsten Crustaceomorphs’
Haug, J. T., Maas, A., & Waloszek, D. (2009). † Henningsmoenicaris scutula,† Sandtorpia vestrogothiensis gen. et sp. nov. and heterochronic events in early crustacean evolution. Earth and Environmental Science Transactions of the Royal Society of Edinburgh, 100(3), 311-350.
Wolfe, J. M., & Hegna, T. A. (2014). Testing the phylogenetic position of Cambrian pancrustacean larval fossils by coding ontogenetic stages. Cladistics, 30(4), 366-390.
Zhang, X. G., Siveter, D. J., Waloszek, D., & Maas, A. (2007). An epipodite-bearing crown-group crustacean from the Lower Cambrian. Nature, 449(7162), 595-598.
Phosphatocopines
Maas, A., Braun, A., Dong, X. P., Donoghue, P. C., Müller, K. J., Olempska, E., … & Waloszek, D. (2006). The ‘Orsten’—more than a Cambrian Konservat-Lagerstätte yielding exceptional preservation. Palaeoworld, 15(3-4), 266-282.
Maas, Andreas & Waloszek, Dieter. (2005). Phosphatocopina – ostracode-like sister group of Eucrustacea. Hydrobiologia. 538. 139-152.
Zhang, X. G., & Pratt, B. R. (2012). The first stalk-eyed phosphatocopine crustacean from the Lower Cambrian of China. Current Biology, 22(22), 2149-2154.
Isoxyiids
Aria, C., & Caron, J. B. (2015). Cephalic and limb anatomy of a new isoxyid from the Burgess Shale and the role of “stem bivalved arthropods” in the disparity of the frontalmost appendage. PloS one, 10(6), e0124979.
Legg, D. A., & Vannier, J. (2013). The affinities of the cosmopolitan arthropod Isoxys and its implications for the origin of arthropods. Lethaia, 46(4), 540-550.
Hymenocarines
Aria, C., & Caron, J. B. (2017). Burgess Shale fossils illustrate the origin of the mandibulate body plan. Nature, 545(7652), 89-92.
Hou, X.., Siveter, D. J., Aldridge, R. J., & Siveter, D. J. (2009). A new arthropod in chain‐like associations from the Chengjiang Lagerstätte (lower Cambrian), Yunnan, China. Palaeontology, 52(4), 951-961.
Moon, J., Caron, J. B., & Gaines, R. R. (2021). Synchrotron imagery of phosphatized eggs in Waptia cf. W. fieldensis from the middle Cambrian (Miaolingian, Wuliuan) Spence Shale of Utah. Journal of Paleontology, 1-12.
Odariids
Izquierdo-López, A., & Caron, J. B. (2019). A possible case of inverted lifestyle in a new bivalved arthropod from the Burgess Shale. Royal Society open science, 6(11), 191350.
Izquierdo‐López, A., & Caron, J. B. (2021). A Burgess Shale mandibulate arthropod with a pygidium: a case of convergent evolution. Papers in Palaeontology, 7(4), 1877-1894.
Legg, D. A., Sutton, M. D., Edgecombe, G. D., & Caron, J. B. (2012). Cambrian bivalved arthropod reveals origin of arthrodization. Proceedings of the Royal Society B: Biological Sciences, 279(1748), 4699-4704.

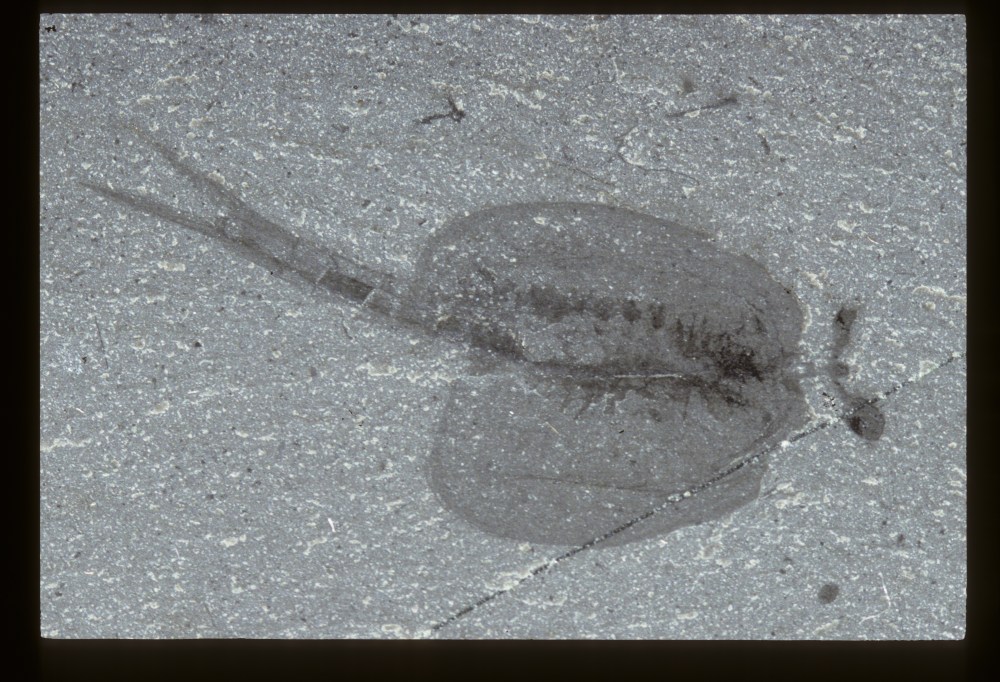


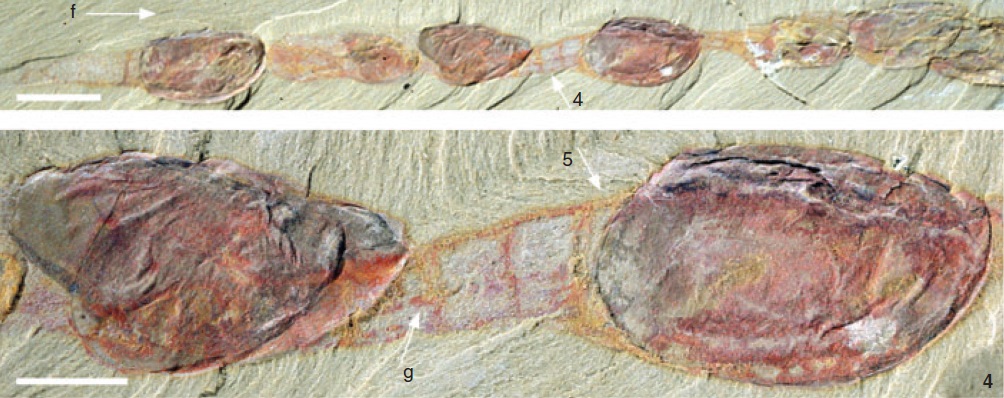

Cool stuff! 🙂
LikeLike